BELOTERO BALANCE (+)
Important Consumer Safety Information
What is BELOTERO BALANCE® (+)?
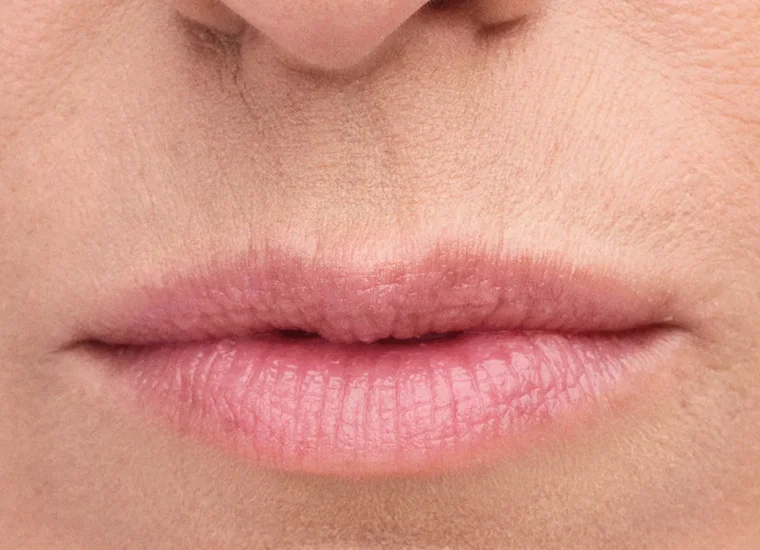
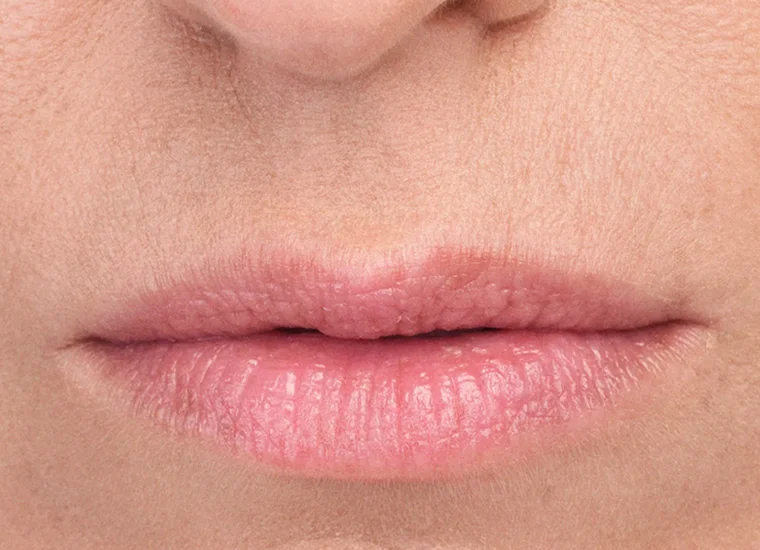
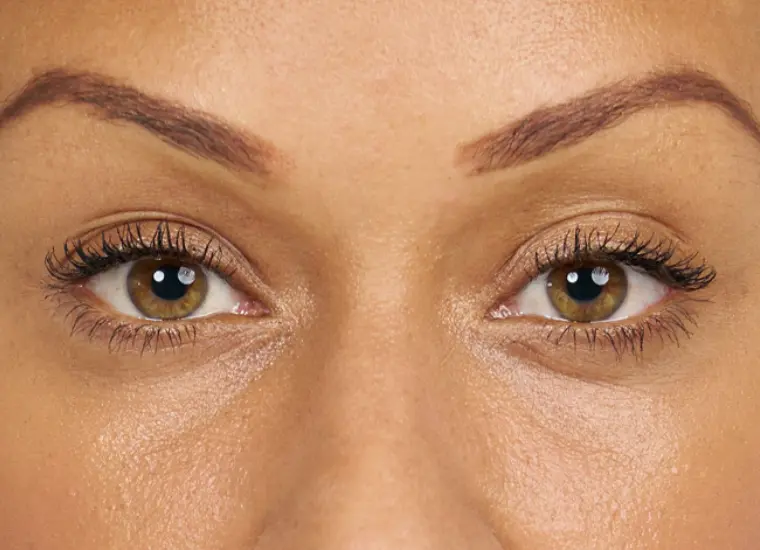
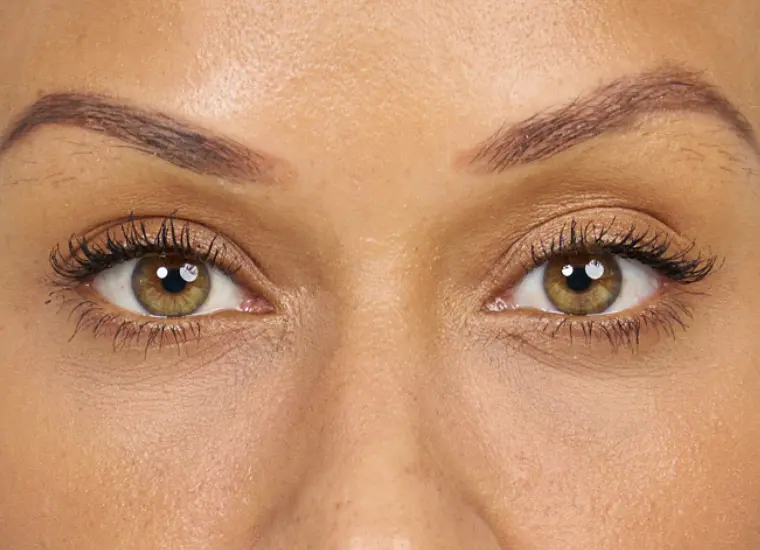
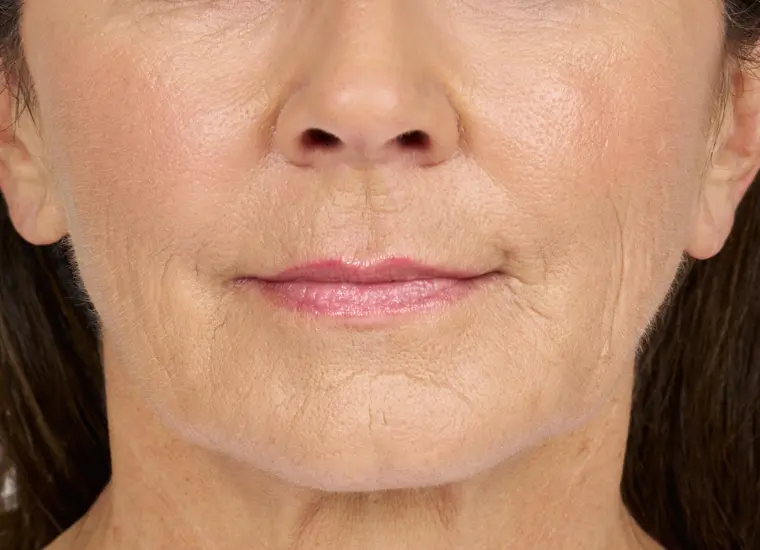
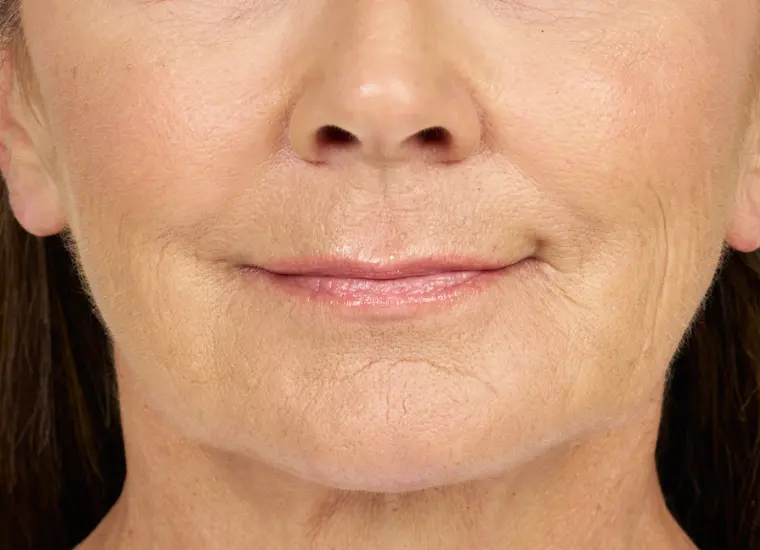
BELOTERO BALANCE is a prescription injection that is approved to temporarily smooth out and fill in moderate to severe nasolabial folds (the folds or wrinkles that go from the side of the nose to the corner of the mouth) and improve the appearance of under-eye hollows in adults over the age of 21.
Who should not use BELOTERO BALANCE?
BELOTERO BALANCE should not be used in patients with a history of or presence of multiple or severe allergies, including those with a history of anaphylaxis. BELOTERO BALANCE should not be used in patients with allergies to gram-positive bacterial proteins.
What is the most important information I should know about BELOTERO BALANCE?
Introduction of BELOTERO BALANCE into the vasculature may lead to embolization, occlusion of the vessels, ischemia, or infarction. Take extra care when injecting soft tissue fillers, for example inject the product slowly and apply the least amount of pressure necessary. Rare but serious adverse events associated with the intravascular injection of soft tissue fillers in the face have been reported and include temporary or permanent vision impairment, blindness, cerebral ischemia or cerebral hemorrhage, leading to stroke, skin necrosis, and damage to underlying facial structures. Immediately stop the injection if patient exhibits any of the following symptoms, including changes in vision, signs of a stroke, blanching of the skin, or unusual pain during or shortly after the procedure. Patients should receive prompt medical attention and possibly evaluation by an appropriate health care practitioner specialist should an intravascular injection occur.
As with all events that involve an injection through the skin, there is a risk of infection. Laser treatments or chemical peels or any other treatments that affect the skin can increase the risk of infection. Do not use BELOTERO BALANCE if you have a skin inflammation or a skin infection. Do not use until the infection is healed.
Patients getting BELOTERO BALANCE may have an injection site reaction. These reactions can include inflammation and usually last less than seven days.
For approximately 24 hours after treatment, avoid:
- strenuous activity
- extensive sun or heat exposure
- aspirin or non-steroidal anti-inflammatory drugs
- alcoholic beverages
Exposure to any of the above can cause temporary redness, swelling, and/or itching at the injection site.
It is not known how BELOTERO BALANCE will work in areas of the face other than the smile lines.
It is not known how BELOTERO BALANCE will work in women who are pregnant or breastfeeding or people who are less than 21 years of age.
What should I tell my doctor before injections with BELOTERO BALANCE?
Tell your doctor if you are taking medicines that affect blood clotting, like aspirin, an NSAID or warfarin. These medicines may put you at an increased risk of bruising or bleeding at the treatment site. Tell your doctor if you have a skin reaction like cold sores, cysts, pimples, rashes, hives, or an infection. Treatment with BELOTERO BALANCE should be delayed until the reaction goes away. Tell your doctor if you are taking medicines that affect your immune system.
What are the most common adverse events seen with BELOTERO BALANCE?
The most common adverse events seen in clinical studies with BELOTERO BALANCE were swelling, bruising, redness, hardening of the skin, pain, altered color, or itching. Other side effects that have occurred in clinical studies of BELOTERO BALANCE include headache, swelling of the side of the nose, moderate cold sore, lip numbness, and lip dryness. Side effects were often mild to moderate and often resolved within 7 days.
Delayed-onset inflammation near the site of injection is one of the known adverse events associated with dermal fillers. Cases of delayed-onset inflammation have been reported to occur at the treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures. Typically, the reported inflammation was responsive to treatment or resolved on its own.
Information on adverse events from post-market surveillance of BELOTERO BALANCE are included in the Package Insert (PI) and Patient Information Guide (PIG) based on an assessment of seriousness and potential causal relationship to BELOTERO BALANCE. Please see the PI and PIG available on www.belotero.com for a complete list of these events.
Important: For full safety information, please visit www.belotero.com or call MyMerz Solutions at 1-844-469-6379 or by email AxUS-adverse.events@merz.com.
Rx only
References: 1. Sundaram H, et al. Plast Reconstr Surg. 2015;136(5 Suppl):149S-163S. 2. Micheels P, et al. Dermatol Surg. 2012;38(7 Pt 2):1162-1169. 3. Tran C, et al. Dermatology. 2014;228(1):47-54. 4. Borrell M, et al. J Cosmet Laser Ther. 2011;13(1):21-27. 5. Belotero Balance (+) [Instructions for Use]. Raleigh, NC: Merz North America, Inc; 2023. 6. Farkas JP, et al. Plast Reconstr Surg Glob Open. 2013;1(1):e8-e15. 7. US Food & Drug Administration. Published July 6, 2023. Accessed October 18, 2023. https://www.fda.gov/medical-devices/aesthetic-cosmetic-devices/dermal-fillers-soft-tissue-fillers 8. Vandeputte J. J Clin Aesthet Dermatol. 2018;11(12);30-39. 9. Merz Aesthetics. Published September 26, 2023. Accessed October 18, 2023. https://us.merzaesthetics.com/news/merz-aesthetics-continues-to-fuel-confidence-by-expanding-belotero-balance-filler-treatment-areas/ 10. Micheels P, et al. J Cosmet Dermatol. 2019;18(1):36-44. 11. Narins RS, et al. Dermatol Surg. 2010;36(s1):730-740. 12. Sieber DA, et al. Plast Reconstr Surg Glob Open. 2016;4(12 Suppl Anatomy and Safety in Cosmetic Medicine: Cosmetic Bootcamp):e1166. 13. American Society of Plastic Surgery. Published 2023. Accessed October 18, 2023. https://www.plasticsurgery.org/cosmetic-procedures/dermal-fillers 14. Aguilera SB, et al. Aesthet Surg J. 2023;43(10):1063-1090. 15. Al-Atif H. Dermatol Pract Concept. 2022;12(1):e2022018. 16. Radiesse [Instructions for Use]. Franksville, WI: Merz North America, Inc; 2023. 17. American Society of Plastic Surgery. Published 2023. Accessed October 2023. https://www.plasticsurgery.org/cosmetic-procedures/dermal-fillers/recovery 18. American Board of Cosmetic Surgery. Published 2023. Accessed October 2023. https://www.americanboardcosmeticsurgery.org/procedure-learning-center/non-surgical/injectable-fillers-guide/ 19. Yale Medicine. Published 2023. Accessed October 2023. https://www.yalemedicine.org/conditions/botox-and-dermal-fillers 20. Brandt FS, et al. Clin Interv Aging. 2008;3(1):153-159.